
Bordwell, private communication.įor a review, see: F. RAMAN Band Correlation Table Approximate Group Intensity Wavenumber Range (cm1) 100210 Lattice vibrations Strong 150430 Xmetal-O Strong 250400 C-C aliphatic chain Strong 295340 Se-Se Strong 425550 S-S Strong 460550 Si-O-Si Strong 490660 C-I Strong. Williams) is available as a PDF file.Ī pKa Values Compilation (by Dave Evans and D. These tables are compiled in PDF files below.įor a more comprehensive discussion on this topic, please see Acidity and Basicity by professor William Reusch, Michigan State University.Ī pKa Values in DMSO Compilation (by Reich and Bordwell) is available as a PDF file.Ī pKa Values in Water Compilation (by R. Below are tables that include determined pKa values for various acids as determined in water, DMSO and in the gas Phase. 
pKa values describe the point where the acid is 50% dissociated (i.e. In short, the stronger the acid, the smaller the pKa value and strong acids have weak conjugate bases. More information on pKa's can be obtained elsewhere (e.g Wikipedia pKa). Typically, organic chemists compare the various values from their determination in water, DMSO and the gas phase and use these to predict a compounds reactivity, solubility, and other physical characteristics. before the suffix and full name of parent alkane is written. If more than one same functional group is present in the compound, then they are indicated as di, tri, etc. The constant changes depending on the solvent the compound is used in. Functional groups like -R, C 6 H 5 -, halogens (F, Cl, Br, I), -NO 2, alkoxy (-OR) etc., are always used as prefix substituents. Its value is directly related to the structure of the given compound. PKa is an acid dissociation constant used to describe the acidity of a particular molecule. pKa Values Compilation (by Dave Evans and D.pKa Values in Water Compilation (by R.pKa Values in DMSO Compilation (by Reich and Bordwell).This is required for students to quickly identify and name the functional groups included in molecules as well as to understand, interpret and draw the specific structure of each functional group clearly. Knowing the functional groups well is one of the fundamental skills required for this course. A summary of common functional groups is included in Table 2.2. 
Up to three hydrocarbon chains can be attached to the oxygen atoms singly bonded to the phosphorus to obtain organophosphates. F unctional groups are the most reactive parts in organic compounds and determine the major properties of compounds. Likewise, a phosphate functional group has a phosphorus atom covalently bonded to four oxygen atoms. 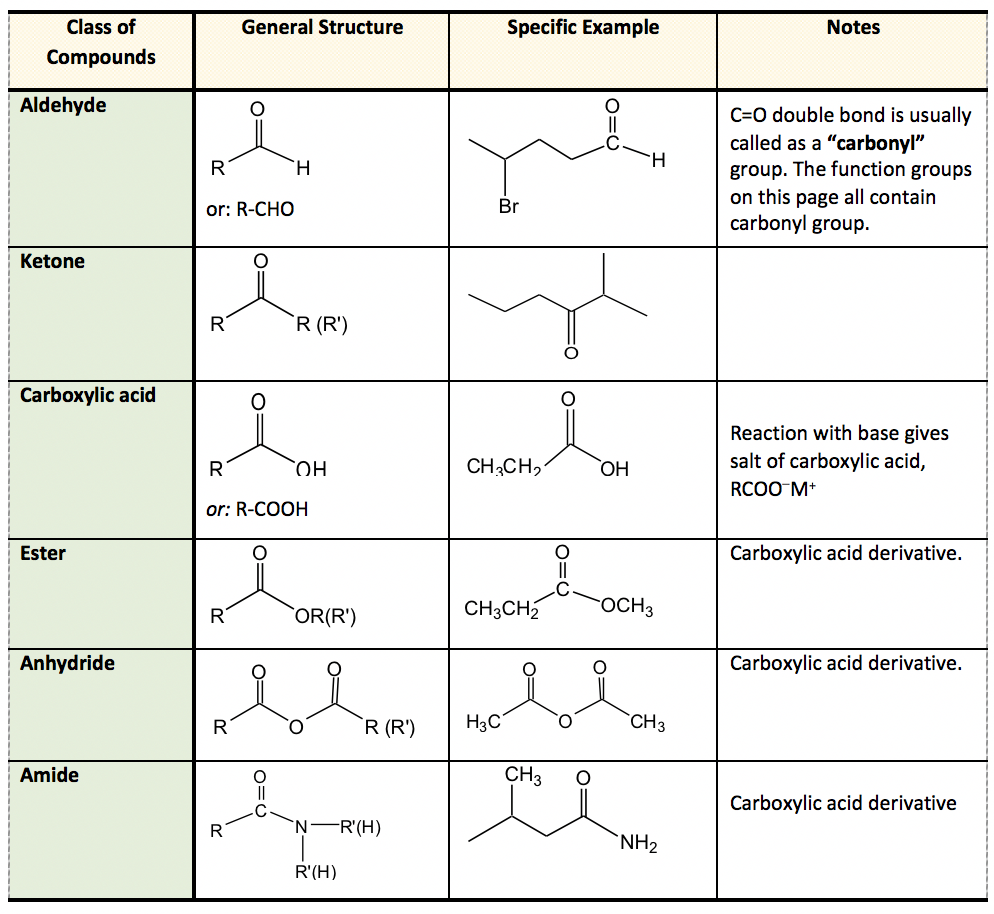
They are classified as primary, secondary, and tertiary, depending on the number of carbon atoms bonded to nitrogen. A hydroxyl group attached to the phenyl-based aromatic ring forms a phenol.Īn amine is a non‑carbon-containing functional group defined by an electronegative nitrogen atom with a lone pair of electrons singly bonded in a hydrocarbon framework. Benzene minus one hydrogen serves as phenyl-a functional group. Multiple bonds are a feature of other functional groups, such as the nitrile or cyano functional group, with a carbon atom triple-bonded to nitrogen.īenzene is an aromatic hydrocarbon that is often represented as six carbon atoms in a ring with alternating single and double bonds. The carbonyl group forms a part of many larger functional groups, such as the carboxyl group of carboxylic acids, which is composed of a hydroxyl group bound to the carbonyl carbon.įunctional derivatives of carboxylic acids include esters, in which the hydrogen of the carboxyl group is replaced by an alkyl group amides, in which the OH of the carboxyl group is replaced by an amine and acyl chlorides, in which the OH is replaced by a chloride. Meanwhile, in ketones, the carbonyl carbon atom is bonded to two other carbon atoms within a hydrocarbon skeleton. The exception is formaldehyde, in which the carbonyl carbon is bonded to two hydrogens. In aldehydes, the carbonyl carbon is bonded to an alkyl group and a hydrogen atom. Two closely related carbonyl-based organic compounds are aldehydes and ketones. One of the important functional groups in organic chemistry is the carbonyl functional group, composed of a carbon atom double-bonded to oxygen. Functional groups are atoms or groups of atoms within an organic compound that impart distinctive chemical properties.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
